SLC26 family
The SLC26 proteins constitute a large family of membrane proteins with diverse functional properties. In humans, the family consists of 11 paralogs that either function as anion exchangers for sulfate, bicarbonate or iodide, “channel-like” uncoupled chloride transporters, or motor proteins, such as Prestin, which is responsible for the contractibility of outer hair cells in the mammalian cochlea. Mutations in SLC26 proteins can lead to several diseases such as Pendred syndrome, chloride-losing diarrhea and deafness.
In the course of our studies, we have determined the structure of a close prokaryotic homologue and characterized its function as H+-coupled fumarate symporter by X-ray crystallography22 and liposome-based functional experiments. The production of specific nanobodies targeting the protein proved essential for obtaining sufficiently diffracting crystals. Current work has shifted from prokaryotic homologues to mammalian SLC26 transporters, which are being characterized by cryo-electron microscopy and electrophysiology. Our research on this protein family is supported by a grant of the Swiss National Science Foundation.
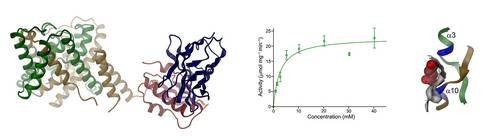
22 Geertsma, E. R. et al. Structure of a prokaryotic fumarate transporter reveals the architecture of the SLC26 family. Nat. Struct. Mol. Biol. 22, 803-808 (2015).