G-protein Coupled Receptors
G-protein coupled receptors
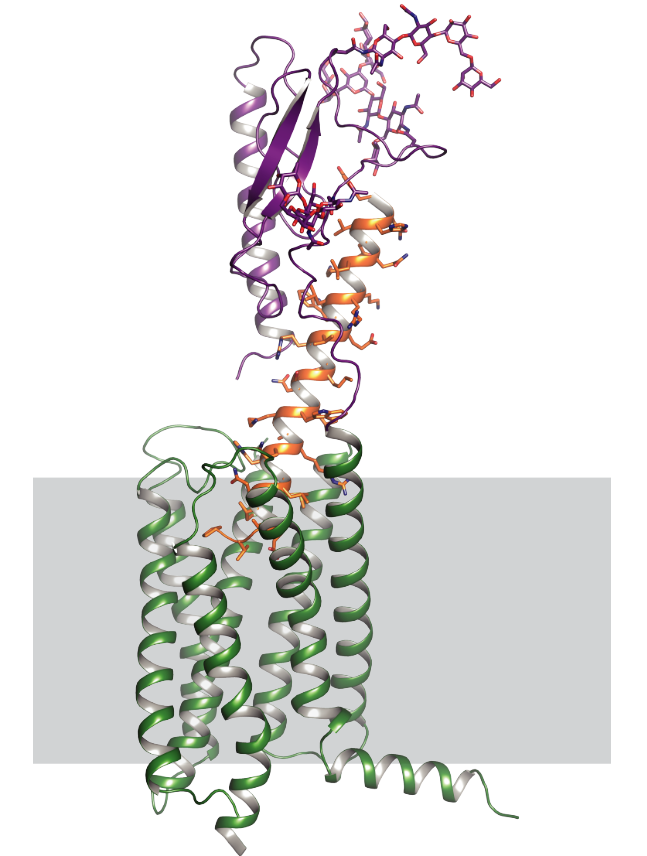
G-protein-coupled receptors regulate a large part of human pharmacology and are the targets of a large fraction of all pharmaceuticals in use today. They undergo characteristic structural changes upon binding antagonists or agonists, and these compounds can be of widely different chemical classes. To understand how specific agonists and antagonists mediate their function, and how to design future active ligands, it is essential to have access to high-resolution structural information. The highest resolution data come from x-ray crystallography, with NMR giving important information on dynamics.
Unfortunately, many wild-type receptors express only in very low yields, are not stable enough to give crystals, or can’t be purified in native form. All of these problems have been solved by a series of protein engineering methods that our laboratory has developed.
This has allowed us to investigate several GPCRs of different families in structural and functional studies, in different states with different ligands, as well as for experimental screening of ligands and antibodies. Among them were the neurotensin receptor, the parathyroid hormone receptor, the neurokinin receptor, the oxytocin receptor, and several others which are currently being worked on.